Empirical studies at the Institute of Biomedical Ethics and History of Medicine (IBME) that do not need mandatory authorization based on the Human Research Act (PDF, 158 KB) (see also the Ordinance (PDF, 152 KB) of the act) will be evaluated internally according to the following procedure. Here, you find all relevant documents and additional information necessary for this evaluation.
Procedure
- Every researcher that plans to execute an empirical study evaluates based on a checklist (PDF, 128 KB) whether the study needs mandatory authorization (see also appendix 1 (PDF, 109 KB)).
- If this is not the case, then the person evaluates using the same checklist (PDF, 128 KB) whether the study has to be evaluated by the internal Review Board (see also appendix 2 (PDF, 66 KB)).
- If the study does not need an internal review, then the person has sole responsibility that the minimal requirements regarding informed consent (PDF, 91 KB), data storage (PDF, 64 KB) and result presentation (PDF, 89 KB) in publications are fulfilled.
Independent from the result of this personal evaluation, the researcher has to hand in a completed copy of the checklist (PDF, 128 KB) to the secretariat of the IBME. The figure below summarizes the procedure.
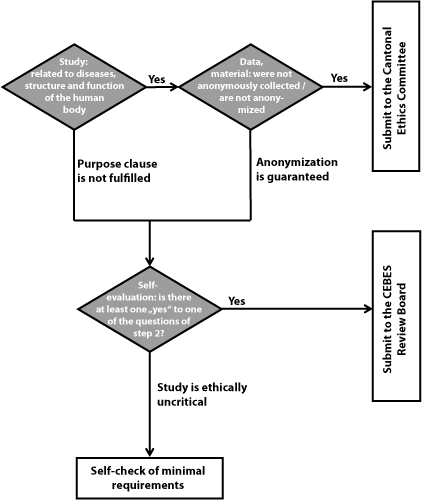
The internal review board consists of the following persons (status: February 2015):
- Roberto Andorno
- Markus Christen
- Tobias Eichinger
- Tanja Krones
Additional information on how the Review Board performs the evaluation is contained in appendix 3 (PDF, 61 KB) and the Review Board Checklist (PDF, 108 KB). For inquiries and submissions to the Review Boards, please use the following e-mail-address: cebes@ibme.uzh.ch
List of all documents:
- Checklist (word (DOCX, 58 KB) / pdf (PDF, 128 KB))
- Appendix 1 (word (DOCX, 30 KB) / pdf (PDF, 109 KB))
- Appendix 2 (word (DOCX, 23 KB) / pdf (PDF, 66 KB))
- Appendix 3 (word (DOCX, 23 KB) / pdf (PDF, 61 KB))
- Informed Consent (word (DOCX, 22 KB) / pdf (PDF, 91 KB))
- Data storage (word (DOCX, 21 KB) / pdf (PDF, 64 KB))
- Presentation of results (word (DOCX, 25 KB) / pdf (PDF, 89 KB))
- Review Board Checklist (word (DOCX, 36 KB) / pdf (PDF, 108 KB))
